The Window of Opportunity in Cognitive Aging
Reframing the Dementia Treatment Problem — Timing, Not Diagnosis
As we have learned from the previous articles Normal vs Non-Normal Brain Aging and Mild Cognitive Impairment – the Gray Zone of Brain Aging, cognitive decline is not defined insofar as its diagnosis but rather its trajectory. The Mild Cognitive Impairment pathway is variable- it may progress towards dementia, resolve or remain a chronic condition.
The variable trajectory reflects a person’s individual state of health – biological, neurological and mental, in recognition that more than one single road may lead to dementia. Multiple, often overlapping domains contribute, including neurological, metabolic, inflammatory, and lifestyle factors.
The Lancet 2024 Commission on Dementia lists the risk factors for developing dementia specifically as lower educational attainment (reduced cognitive reserve), hearing loss, hypertension, smoking, obesity, depression, diabetes, physical inactivity, alcohol consumption, traumatic brain injury (TBI), air pollution, social isolation, vision loss and elevated LDL cholesterol levels. 1
Within the clinical context of preemptive and rehabilitative treatment for MCI and dementia one critical question emerges. Is there still time for effective intervention?
When is it too late? Is there still time for effective intervention?
The answer depends on whether the biological imbalances remain modifiable and secondly, on the individual’s willingness to engage in meaningful change.
The article will discuss and map out the phases of brain aging, cognitive decline, and neurodegeneration as they progress toward dementia; the role of cognitive reserve; the evidence that the trajectory can be influenced; and ultimately, the “window of opportunity” as a dynamic concept guiding effective clinical intervention.
Phases of Brain Aging versus Awaiting the Dementia Diagnosis
Abundant research demonstrates that MCI and dementia develop over years, if not decades. Importantly, clinical indicators such as amyloid-β and tau proteinopathies are increasingly understood as downstream effects of processes driving neurodegeneration, rather than primary causes.
Identifying upstream causality is critical; it allows us to open the window and examine the biological phases of brain aging and in turn, identify the periods of greatest therapeutic opportunity.
Before preceding further, several guiding principles frame this perspective on brain aging and neurodegeneration.
- While Mild Cognitive Impairment is seen as a transitional stage between cognitive decline of health aging and dementia, there is a significant element of variability in its trajectory.2
- Functional decline in neurological networks as a result of synaptic loss precedes clear structural correlation.2
- Early inflammatory3, vascular4, metabolic5,6, mitochondrial5,6 and glymphatic7,8 dysfunctions precede dementia onset and may be seen as upstream factors in a gradual early phase neurodegenerative process.
- Early phase involvement refers to upstream functional / metabolic processes and is largely modifiable.
- Late phase refers to overt pathology diagnosis i.e. dementia and involves structural degenerative changes.
- Individuals may present with single or multiple upstream pathologies.
- Best practice therapeutic methods are determined by the individual health circumstances. There is no fixed boundary – the therapeutic process is dynamic and individual.
Taken together, these principles shift the clinical lens from diagnosis to timing.
A diagnosis such as MCI or dementia represents a late-stage categorization within a much longer biological process. Therefore, understanding and addressing the upstream phases of brain aging aligns clinical intervention with underlying biology- at a point where functional change may still be stabilized or improved.
The “window of opportunity” then emerges not as a fixed period, but as an individualized function of ongoing biological modifiability.
What Drives Dementia Trajectory Forward - Mechanisms of Degenerative Acceleration
In the case of obesity, no individual wakes up one morning and decides, “Tomorrow I will become obese.” It is not an abrupt event, but a gradual process—one that develops over time, often accompanied by the belief that the future will somehow look different, without meaningful change in behavior.
The same principle applies to dementia.
Cognitive decline does not emerge suddenly. It evolves through the cumulative influence of multiple interacting factors over years, often decades. This creates a false sense of reassurance, especially when individuals insist they do not meet the classic or overt risk profiles.
“I exercise, I don’t smoke, I don’t have diabetes—therefore I won’t develop dementia.”
Not so fast.
The central issue is not the presence or absence of individual risk factors. The salient feature is rather the multiplicity of processes and their dynamic interaction over time. As highlighted in the 2024 Lancet Commission on Dementia, risk is cumulative, and importantly, it is not limited to overt disease states.
Subclinical dysfunction such as metabolic, vascular, inflammatory, and neurological may be present long before it crosses a diagnostic threshold. Most importantly, the absence of overt pathology does not imply the absence of underlying processes.
These processes may exist:
- at diagnostic levels
- or within a sub-diagnostic, “border-range” state
Yet in both cases, they act as drivers of degenerative acceleration.
These processes do not operate in isolation. They interact and reinforce one another, over time accelerating a steep descent in brain aging.
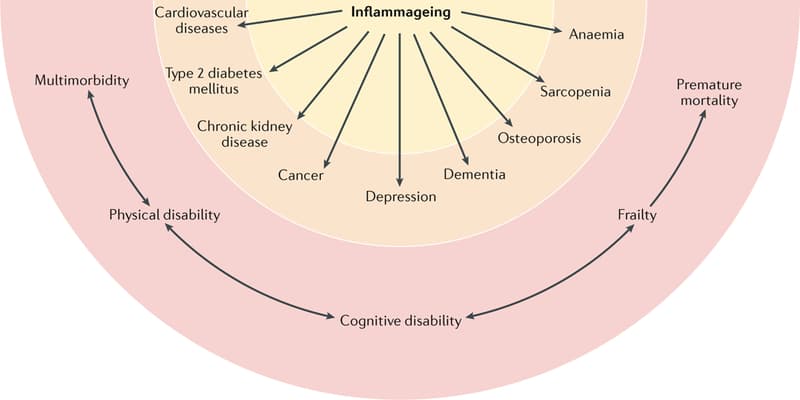
Inflammaging – Systemic Inflammatory Pressure
Chronic low-grade inflammation develops gradually over the lifespan and is a central driver for degenerative biological aging. This persistent state is reinforced by obesity, gut permeability, gut microbiome imbalances, mitochondria dysfunction, immune dysregulation, chronic infections and environmental exposures.3 These processes remain subclinical for years while maintaining an internal environment of biological instability.
Over time, this inflammatory burden contributes to widespread systemic effects, including cardiovascular disease, diabetes and metabolic dysfunction, sarcopenia, depression and neurodegeneration-dementia. 3
Within the brain, these changes disrupt vascular integrity, neuronal signaling, and metabolic stability, contributing to progressive network dysfunction well before structural degeneration becomes apparent.
Ferrucci L, Fabbri E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol. 2018;15(9):505–522.
Vascular Dysfunction & Blood Brain Barrier Compromise – Delivery System Breakdown
Vascular integrity plays a central role in maintaining brain health, serving as the primary system for nutrient delivery, waste exchange and regulation of the neural environment. Over time, structural and functional changes within the vasculature such as arterial stiffening, impaired cerebrovascular reactivity, and microvascular injury begin to alter cerebral blood flow dynamics.4
These changes often remain subclinical in early stages but progressively affect the blood brain barrier stability. Reduced barrier integrity, leakages and microbleeds have been observed in early phases of dementia, including within vulnerable regions such as the hippocampus. 4
As vascular function deteriorates, the brain becomes increasingly exposed to inflammatory mediators, impaired nutrient delivery and reduced metabolic support. Over time, this contributes to the accumulation of pathological processes amyloid β plaque burden, while simultaneously destabilizing neural networks. 4
The net effect is a progressive breakdown in the brain’s delivery and regulatory systems, reducing the brain’s tolerance for dysfunction and leading towards a more vulnerable trajectory of decline.
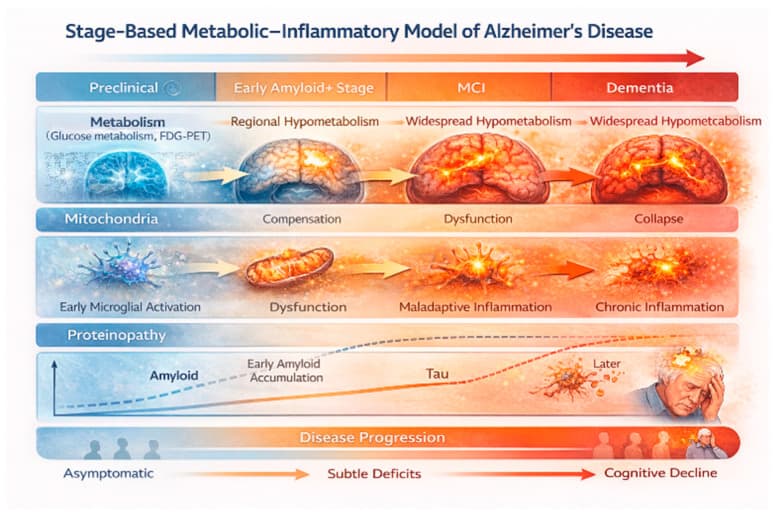
Metabolic Dysfunction & Glucose Dysregulation – Cellular Energy Failure
Metabolic and inflammatory imbalances are early central contributors to cognitive decline and dementia. Insulin resistance and insufficiencies in brain glucose metabolism lead to reduced energy utilization, reduced brain metabolic activity and altered regulation of amyloid β production and clearance.5,6
Reduced glucose availability makes neurons and synapses more susceptible to damage, resulting in lower activity as well as increased production and impaired clearance of pathological proteins. Insulin resistance further contributes to pathological change through increased tau phosphorylation.
Mitochondrial dysfunction leads to reduced ATP levels, lower cellular energy availability and increased reactive oxygen species, creating a state of oxidative stress that contributes to neuronal damage, synaptic failure and accumulation of amyloid β and tau. 5,6
FDG-PET observations of reduced cerebral glucose hypometabolism in temporoparietal and posterior cingulate regions correlate more strongly with cognitive decline than plaque burden, reinforcing the role of metabolic dysfunction as an early feature rather than a downstream effect.5
Christodoulou RC, Eller D, Papageorgiou PS, et al. Metabolic Dysfunction in Alzheimer's Disease: Brain Glucose Hypometabolism as an Early Precursor to Amyloid and Tau Pathology. J Clin Med. 2026;15(5):1884.
Neuroinflammation is a stage-dependent process linked to metabolic dysfunction. Early phases involve microglial activation associated with metabolic hyperactivity, while later phases progress toward hypometabolism, reflecting reduced cellular activity and eventual neurodegeneration.5
Cerebral hypometabolism therefore represents an early indicator of neurodegenerative progression, often preceding overt structural change.
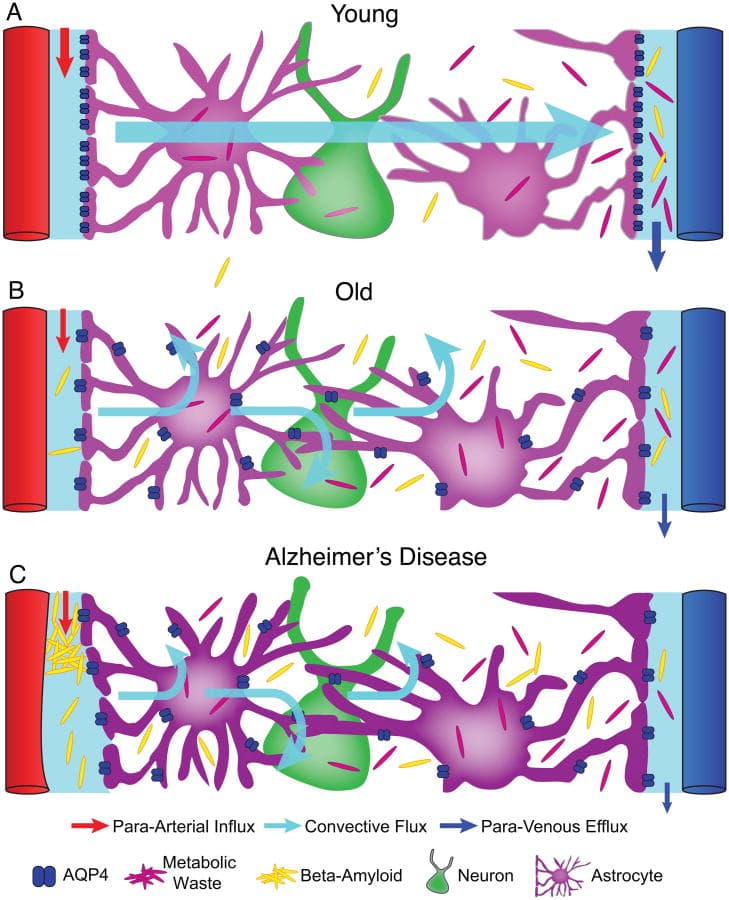
The Glymphatic System – Clearance System Failure
Optimal waste clearance is essential for maintaining brain homeostasis. The glymphatic system plays a central role in the clearance of metabolic waste products, including amyloid β and tau. This recently characterized system relies on the movement of cerebrospinal fluid (CSF) through perivascular spaces, facilitating exchange with interstitial fluid and enabling removal of neurotoxic byproducts generated during normal neuronal activity.
Glymphatic function is closely coupled to sleep architecture, vascular pulsatility and astroglial integrity. In fact, it functions primarily during sleep and remains generally “deactivated” during wakefulness. Disruptions such as sleep fragmentation, reduced slow-wave sleep, vascular stiffness and astrocytic dysfunction may impair the efficiency of fluid exchange and metabolic clearance. These changes often develop gradually and may remain subclinical for extended periods.7,8
With age, glymphatic function becomes less effective and metabolic waste begins to accumulate within the interstitial space. In the context of cognitive aging and dementia, impaired clearance contributes to the buildup of amyloid β, tau, inflammatory mediators, and other neurotoxic byproducts, further exacerbating neuroinflammatory signaling. A vicious cycle emerges, perpetuating impaired clearance and increasing pathological burden. Additionally, the glymphatic role in maintaining ionic and nutrient balance across brain tissue becomes increasingly disrupted. 7,8
The net effect is a progressive failure of the brain’s waste clearance systems which reduces neurological tolerance for metabolic burden and increases susceptibility to ongoing degeneration.
Jessen NA, Munk AS, Lundgaard I, et al. The Glymphatic System: A Beginner’s Guide. Neurochem Res. 2015;40(12):2583–2599.
The progressive accumulation of these different factors is a reduced neurological tolerance for dysfunction. As understood across inflammatory, vascular, metabolic, and glymphatic-clearance related functions, the homeostatic failures driving neurodegeneration exist as upstream biological processes years, if not decades, prior to the emergence of cognitive symptoms. Over time, these dysfunctions contribute to middle-stage network disturbances and later-stage structural degeneration associated with more significant loss of cognitive function.
The critical takeaway is there is no fixed boundary separating these phases; rather, the changes are dynamic, cumulative and highly individualized.
Cognitive Reserve — Brain Resilience in Cognitive Aging and Dementia
Individuals with higher lifelong educational and occupational attainment tend to have a reduced risk for developing Alzheimer’s disease and dementia. This observation is referred to as Cognitive Reserve (CR) and reflects an overall state of greater brain resilience to age-related brain changes and Alzheimer’s disease.
Brain Reserve, or passive reserve, refers to having more neurons and synaptic connections available to lose. In other words, a larger brain. In this instance, a greater degree of pathological burden can be tolerated before a critical degenerative threshold is reached and symptoms begin to emerge.9
Cognitive Reserve, an active reserve or process, refers to a greater degree of functional brain activity and adaptability, thereby facilitating more effective coping with damage through neurological compensation before a critical threshold is met. In CR the level of functionality is the primary element, irrespective of brain size. Life experience has also been suggested to reduce susceptibility to age-related brain pathology.9
Based on the concept that pathology accumulates across a timeline separately from Cognitive Reserve, it would take longer for an individual with higher reserve CR to reach a point of clinical dementia diagnosis. Studies demonstrate that individuals with low educational attainment (8 years) or low occupational attainment carry approximately a 2–2.25 times greater risk of developing dementia compared with individuals of higher attainment. Likewise, individuals engaged in 6 or more leisure activities such as walking, music, visiting friends, reading, volunteering, and attending church or synagogue, demonstrated a 38% reduced risk of developing dementia.9
Strikingly, individuals with high Cognitive Reserve often demonstrate a more rapid decline once diagnosed with Alzheimer’s disease compared with lower reserve individuals. PET imaging corroborates this observation, demonstrating more severe Alzheimer’s disease-related pathological patterns in individuals with higher educational attainment. One explanation is that the degenerative process has been functionally masked for longer and has therefore reached a more advanced stage by the time compensatory resilience begins to fail.9
Reserve is not simply protective; it is also modifiable. Later-life interventions have been shown to increase Cognitive Reserve, slow age-related decline and support healthier aging trajectories. Complex gameplay and focused cognitive training have demonstrated improvements across multiple cognitive domains, although training within one domain does not necessarily positive impact others.9
Aerobic exercise in individuals with below-average respiratory capacity has demonstrated improvements in cognitive performance. Furthermore, resistance-based strength training has also been demonstrated to improve cerebrovascular function and overall brain health in aging.10 Longitudinal findings from the IDEAL study further suggest that Cognitive Reserve continues to influence cognitive function, quality of life and functional ability even following a diagnosis of dementia.11
An exact “recipe” for determining best outcomes has yet to be established in the research. However, reserve it is best understood as highly individualized and influenced by the multiplicity of factors so far addressed in this article.
The STAC Model — How the Brain Compensates
Structural changes typical of the aging brain include reductions in brain volume, loss of white matter integrity and depletion of dopamine receptors. Structural shrinkage is particularly observed in regions such as the caudate, cerebellum, hippocampus, and prefrontal cortex. Reduced white matter integrity is most significant within frontal brain regions and is associated with demyelination, inflammatory processes, trauma and other forms of neural injury. Dopamine receptors likewise play an integral role in attention regulation and response modulation to contextual stimuli.12
Cognitive decline develops in parallel with processes driving reductions in brain volume, white matter integrity, and dopamine receptor availability. However, direct relationships between structural changes and cognitive symptoms are not always consistently observed. Of the structural changes, white matter integrity appears to demonstrate a stronger relationship with cognitive function than overall brain volume alone. 12
The major question bridging cognitive neuroscience and gerontology is what accounts for the high cognitive performance seen in advanced old age, even with various coexisting pathologies.
Increases in functional brain activity, especially in the frontal cortex, are observed at the same time of age-related structural brain and cognitive changes. The Scaffolding Theory of Aging and Cognition (STAC) describes this compensatory recruitment of neural networks in response to inefficient, noisy, or declining neural processing. This compensatory neuroplastic behavior predominates within the prefrontal cortex because of its inherent flexibility and adaptive capacity.
In essence, there is a shifting locus of function — a scaffolding of compensatory activity into the prefrontal cortex to offset inefficiencies in parahippocampal and mediotemporal activation.
To illustrate, adaptive scaffolding responses during cognitive task demand are observed between younger and older adults. Younger adults recruit both right and left prefrontal regions for high level task demand and unilaterally for low level task demand. Older adults demonstrate more consistent bilateral prefrontal recruitment even during lower-level cognitive task demand, reflecting greater reliance on compensatory network activation.
Scaffolding is not unique to aging; it reflects a broader adaptive property of the brain in response to cognitive challenge. Aging, however, increases the frequency and magnitude of these challenges and drives greater reliance on compensatory network recruitment. Scaffolded networks are generally less efficient than the networks they compensate for and effective scaffolding declines with both aging and neurodegenerative pathology. 12
Importantly, cognitive activity itself appears to stimulate scaffolding processes, reinforcing the concept that compensatory brain function remains dynamic and potentially modifiable across the aging process.
The ACTIVE STUDY – Evidence that Cognitive and Functional Independence Can Be Improved
While numerous studies have demonstrated that systematic cognitive training interventions can improve cognitive performance in older adults, the ACTIVE Study sought to determine whether cognitive training could translate into improvements in real-world functional independence. In other words, could cognitive training transfer into meaningful real world everyday activities and outcomes in old age?
Memory, reasoning and processing speed were selected for training as these domains tend to decline earlier with age (mid 60s) compared to other cognitive functions. These domains are particularly salient because of their high level of requirement in activities of daily living and independent living. The driving premise of the ACTIVE Study was that targeted domain training would transfer to real world activities associated with those domains, and in turn, would positively influence overall quality of life and functional independence.13
It is critical to note, of the 2,802 individuals recruited, the participants did not have significant cognitive impairment at baseline. Rather, they represented an aging population at risk for cognitive decline because of elevated risk factors such as hypertension and diabetes. The average participant was 74 years old and the study included both men and women, Caucasian and African American populations.
Memory training focused on verbal episodic memory. Reasoning training focused on serial pattern problem solving, while speed training emphasized visual search and the ability to process and inspect increasing amounts of information over shorter time periods. Participants completed 10 training sessions lasting 60-75 minutes over the course of 5-6 weeks. Booster training sessions of 4 additional sessions were performed later at 11 and 35 months.
How would this training relate to real life and what were the findings?
Activities including meal preparation, housework, financial management, healthcare management, telephone use, shopping, dressing, personal hygiene, information search and retrieval, medication label interpretation, grocery lists and driving were assessed. Each intervention training demonstrated immediate improvement with measurable durability at the time of follow up 5 years later! The greatest improvement seen was in the processing speed followed by reasoning and memory. 13
The broader implication of this landmark study is that cognitive trajectory is modifiable not fixed. Targeted cognitive intervention can influence both cognitive performance and functional independence later in life.
Clinical Implications — From Detection to Meaningful Redirection
Is the Window of Opportunity being missed? Waiting for symptom-based detection of cognitive change would certainly imply that it is. Hyperfocus on structurally driven models of detection, rather than utilizing functional and subclinical markers relating to inflammatory, vascular, metabolic and glymphatic dysfunction for example, neglects critical periods during which precursors to network and structural loss are already developing. The underlying biological processes have often been active for years or decades — why wait to intervene?
The critical clinical question therefore is not simply whether an individual meets criteria for Mild Cognitive Impairment or dementia, rather
- Which direction is the cognitive trajectory moving towards?
- What are the biological, behavioral and environmental drivers?
- To what degree are the drivers modifiable?
- Has a critical threshold been reached or exceeded?
- Do subthreshold features remain sufficiently modifiable to help compensate for other functions already lost?
- What degree of willingness for time intensive intervention do the individual and relevant family members embrace?
Longitudinal assessment and intervention therefore become essential because cognitive decline is not a static event, but a dynamic process unfolding across time.
Timing Is the Intervention
Cognitive trajectory is dynamic, reserve remains modifiable and risk is influenceable. Outcomes are therefore not entirely predetermined.
The window of opportunity is not a single moment in time. It is a biological phase determined by multiple interacting variables during which dysfunction remains sufficiently modifiable and future trajectory can still be influenced. As underlying pathological burden accumulates, that window progressively narrows.
Timing therefore becomes one of the most important interventions in cognitive aging and dementia care. These principles form the foundation of the Empowered Brain Program, our longitudinal neurorehabilitation approach focuses on identifying and addressing the biological, behavioral, and environmental factors influencing cognitive trajectory and functional decline.
- Livingston G, Huntley J, Liu KY, et al. Dementia prevention, intervention, and care: 2024 report of the Lancet standing Commission. Lancet. 2024;404(10452):572-628. doi:10.1016/S0140-6736(24)01296-0
- Harada CN, Natelson Love MC, Triebel KL. Normal cognitive aging. Clin Geriatr Med. 2013;29(4):737-752. doi:10.1016/j.cger.2013.07.002
- Sweeney MD et al. Vascular dysfunction-The disregarded partner of Alzheimer's disease. Alzheimers Dement. 2019 Jan;15(1):158-167. doi: 10.1016/j.jalz.2018.07.222. Erratum in: Alzheimers Dement. 2022 Mar;18(3):522. doi: 10.1002/alz.12483.
- Ferrucci L, Fabbri E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol. 2018 Sep;15(9):505-522. doi: 10.1038/s41569-018-0064-2.
- Christodoulou RC, Eller D, Papageorgiou PS, Angelopoulou E, Vassiliou E, Papageorgiou SG. Metabolic Dysfunction in Alzheimer's Disease: Brain Glucose Hypometabolism as an Early Precursor to Amyloid and Tau Pathology. J Clin Med. 2026 Mar 1;15(5):1884. doi: 10.3390/jcm15051884.
- Dewanjee S, Chakraborty P, Bhattacharya H, Chacko L, Singh B, Chaudhary A, Javvaji K, Pradhan SR, Vallamkondu J, Dey A, Kalra RS, Jha NK, Jha SK, Reddy PH, Kandimalla R. Altered glucose metabolism in Alzheimer's disease: Role of mitochondrial dysfunction and oxidative stress. Free Radic Biol Med. 2022 Nov 20;193(Pt 1):134-157. doi: 10.1016/j.freeradbiomed.2022.09.032. Epub 2022 Oct 4.
- Buccellato FR, D'Anca M, Serpente M, Arighi A, Galimberti D. The Role of Glymphatic System in Alzheimer's and Parkinson's Disease Pathogenesis. Biomedicines. 2022 Sep 13;10(9):2261. doi: 10.3390/biomedicines10092261.
- Jessen NA, Munk AS, Lundgaard I, Nedergaard M. The Glymphatic System: A Beginner's Guide. Neurochem Res. 2015 Dec;40(12):2583-99. doi: 10.1007/s11064-015-1581-6. Epub 2015 May 7.
- Stern Y. Cognitive reserve in ageing and Alzheimer's disease. Lancet Neurol. 2012 Nov;11(11):1006-12. doi: 10.1016/S1474-4422(12)70191-6.
- Allison EY, Bedi AM, Rourke AJ, Mizzi V, Walsh JJ, Heisz JJ, Al-Khazraji BK. Resisting decline: the neuroprotective role of resistance exercise in supporting cerebrovascular function and brain health in aging. Front Physiol. 2025 Sep 22;16:1606267. doi: 10.3389/fphys.2025.1606267.
- Gamble LD, Clare L, Opdebeeck C, Martyr A, Jones RW, Rusted JM, Pentecost C, Thom JM, Matthews FE. Cognitive reserve and its impact on cognitive and functional abilities, physical activity and quality of life following a diagnosis of dementia: longitudinal findings from the Improving the experience of Dementia and Enhancing Active Life (IDEAL) study. Age Ageing. 2025 Jan 6;54(1):afae284. doi: 10.1093/ageing/afae284.
- Park DC, Reuter-Lorenz P. The adaptive brain: aging and neurocognitive scaffolding. Annu Rev Psychol. 2009;60:173-96. doi: 10.1146/annurev.psych.59.103006.093656.
- Tennstedt SL, Unverzagt FW. The ACTIVE study: study overview and major findings. J Aging Health. 2013 Dec;25(8 Suppl):3S-20S. doi: 10.1177/0898264313518133.